
Here is a simple mnemonic to memorize these six acids 'Glaciers in Alaska valiantly locate isolated prowlers'. They are nonpolar and therefore hydrophobic. Lets begin with a group of aliphatic amino acids.
#HYDROPHOBIC AMINO ACIDS MNEMONIC ISO#
They will have iso electric points at a pH lower than physiological pH and be negatively charged at physiological pH (You need a more acidic environment to obtain an overall neutral charge because the carboxyl groups can more easily lose a proton). Amino Acid Mnemonic Tricks for Easy Name Memorizing. Meaning for Non-polar (hydrophobic) amino acids. Similarly, amino acids that are said to be acidic have an extra carboxyl group. Study Amino acids flashcards from Dylan Halls Roseman University of Health Sciences class online. In other words, their pKa’s are high enough that they tend to bind protons, gaining a positive charge in the process. External www files: Mnemonics to help you remember 1-letter codes Amino acid explorer. Aromatic rings make bulky side chains which may affect coiling of the polypeptide chain. (hydrophobic), so they may face to centre of molecule. They will have iso electric points at a pH higher than physiological pH and will be positively charged at physiological pH (You need a more alkaline environment to obtain an overall neutral charge because the amino group can hold onto another proton). Amino acid Click to bring up 3-D representation (separate window) 3-letter and 1 letter. I was talking about being positive in the physiological pKa.Īll amino acids can act as either an acid or a base because they have a carboxyl group than can lose protons and an amino group than can gain protons/donate electrons.Īmino acids that are said to be basic have an extra moiety which will accept electrons (Their side chains contain nitrogen and resemble ammonia, which is a base at neutral pH). Hey don’t get confused between your “basic” concepts! *pun intended* caused by the repulsion between departing proton and nearby positively charged amino group on the alpha carbon -pKa of the amino group in glycine is perturbed downward relative to the average pKa of an amino group due to partly electronegative O2 atoms in carboxyl groups, which tend to pull electrons toward them, increasing the tendency of the amino group to give up a proton. Acidic, there’s only 2 Glutamate, Aspartate. TCSy-TAG like cyborg, charged Tyrosine, Cysteine, Serine, Threonine, Asparagine, Glutamine. Wait, why would they be positive again? Aaaaahhhh GLAMP TVIP nonpolar, hydrophobic Glycine, Leucine, Alanine, Methionine, Phenylalanine, Tryptophan, Valine, Isoleucine, Proline. Should you wish to submit your own content, please consider buying a sponsored link from Reddit.Basicity: the willingness to donate electrons Please do not post screenshots of copyrighted material.Ĭan I share my blog/website/business? This subreddit is not a place to spam your blog or solicit business.

This includes discussion of filesharing or sources of pirated materials Political posts are not allowed unless explicitly related to premed.įilesharing is prohibited in this subreddit.
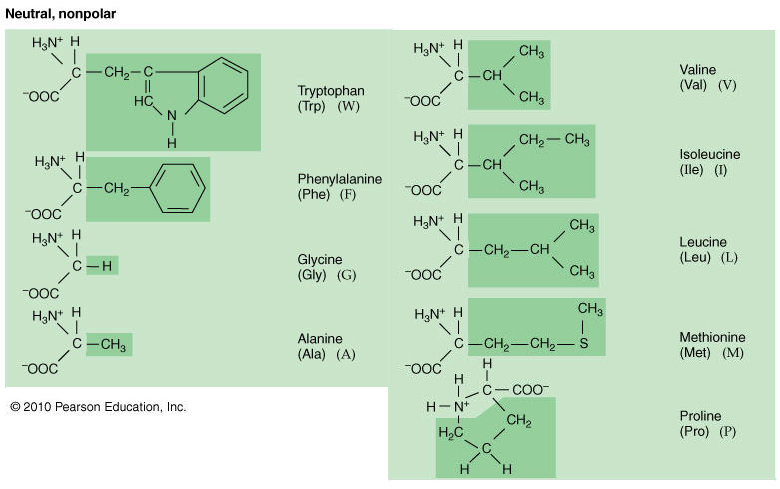
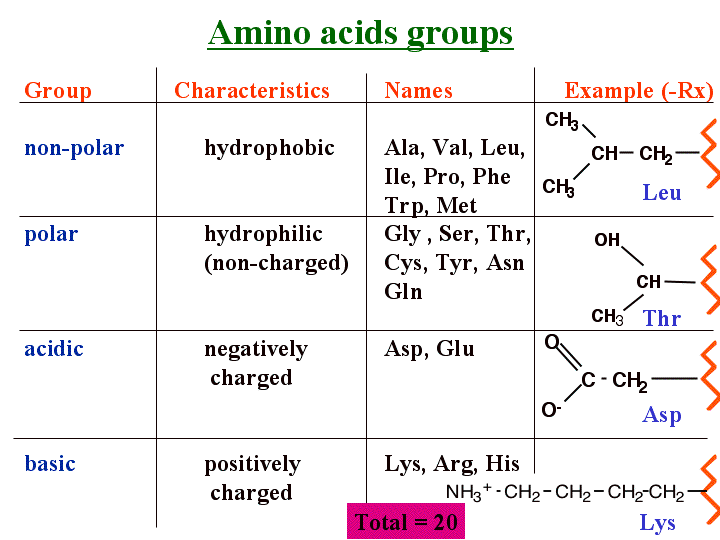
Please post only on-topic MCAT posts or questions. In order to form a hydrogen bond with water, a polar molecule, the amino acid side chains must also be polar, or have an unequal distribution of electrons. Please see /r/premed if you have questions regarding the premedical curriculum and/or applying to medical school. Hydrophobic amino acids are composed primarily of carbon atoms, which cannot form hydrogen bonds with water. Any form of racism, sexism, homophobia, or general unnecessary extremely offensive behavior will not be tolerated.ĭoxxing or revealing of personal information of any kind is not allowed. Please be respectful to your fellow posters.
